- Home
-
Clinical Solutions
Interventional AccessHemostasis
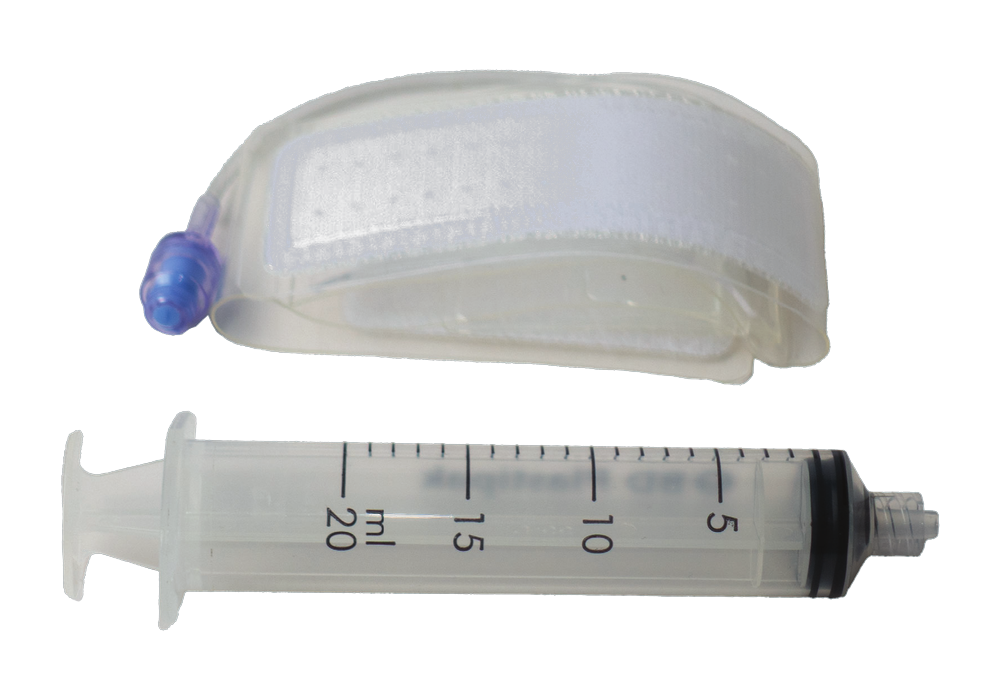 Trans Radial Band
Bone Cement
Trans Radial Band
Bone Cement - Procedure Packs
- Quality & Compliance
- Contact
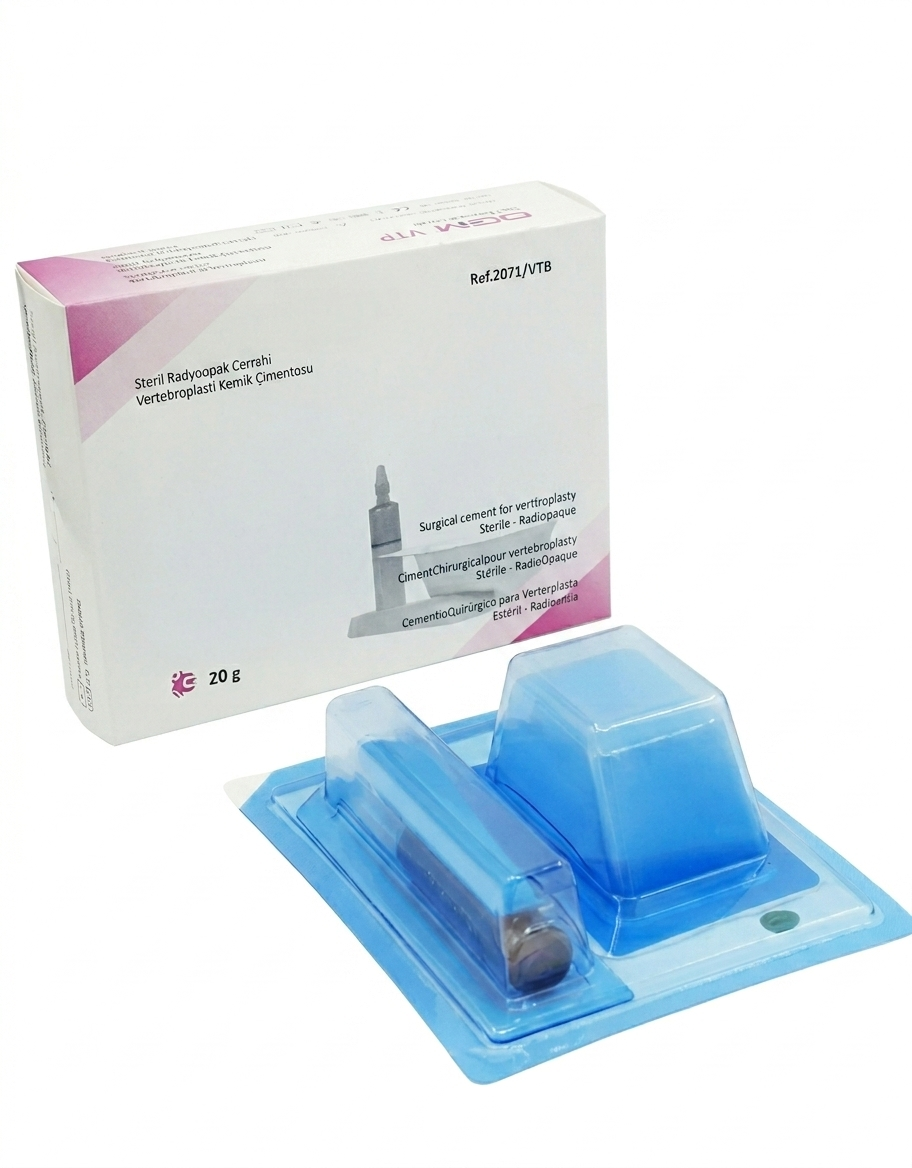
PMMA-based radiopaque surgical cement specifically formulated for vertebroplasty procedures. Stabilizes fractured, collapsed, or compressed vertebral bodies caused by osteoporosis or pathological conditions such as tumors.
An acrylic-based, radiopaque surgical bone cement containing PMMA (polymethyl methacrylate), formulated specifically for vertebroplasty. Indicated for the consolidation and stabilization of fractured, collapsed, or compressed vertebral bodies caused by osteoporotic or pathological conditions including tumors.
Engineered with a low reaction temperature to minimize exothermic damage to surrounding bone and soft tissue. Its high radiopaque content provides excellent fluoroscopic visibility throughout the injection procedure. Supplied as a two-component system — sterile powder and sterile liquid monomer in adjacent ready-to-use blisters, no external mixing tools required.
| Reference | Product | Pack size |
|---|---|---|
| 0308/VTB | Vertebroplasty Cement | 12.5 g |
| 2023/VTB | Vertebroplasty Cement | 15 g |
| 2071/VTB | Vertebroplasty Cement | 20 g |
| 1406/VTB | Vertebroplasty Cement | 25 g |
Request a quote, a sample, or technical details — our team responds within one business day.
Contact Us